 username@email.com
username@email.com
In this lesson we will review chemical names and formulas for acids. We will also review the acid/base properties of oxides.
Although there are multiple theories regarding what constitutes an acid and a base, the two theories most commonly used in the chemistry classroom and laboratory are the Arrhenius theory and the Bronstedt-Lowry theory. They agree on this point: an acid must contain hydrogen ions, H+. Because acids are compounds and therefore must be electrically neutral, they are required to have an anion with them.
Remembering this makes writing names and formulas for acids relatively simple, in that all acids will contain the same cation—H+—and will differ only by the anion. Thus, we merely need to learn the relationship between the anion name and the acid name.
Binary acids all have anions whose names end with –ide. For example, the binary acid HCl is made from the chloride ion, Cl1−. Its formula is written like that of any other ionic compound. We need one H+ ion and one Cl1− ion for the charges to add up to zero.
We name the acid by using the following pattern. We must drop the –ide ending from the anion name, in this case turning chloride into chlor. We take the root that remains (chlor), place the prefix hydro in front of it, the suffix –ic after it, and the word acid at the end. In this way, the name chloride turns into hydrochloric acid.
We can reverse this process to determine the formula of a binary acid if given the name. Hydroiodic acid displays the hydro-_____-ic acid pattern of a binary acid; therefore, its anion name must end with –ide. From this, we realize that the ions involved are hydrogen (H+) and iodide (I1−), so the formula must be HI.
It is worth mentioning that there is one acid which is not binary but still follows this pattern. HCN contains three different elements, but the anion name is cyanide; therefore, the acid name is hydrocyanic acid.
Which of the following is the correct name for HBr?
The correct answer is D. Neither choice A nor B uses the prefix hydro. Choice C has an incorrect suffix (–ous rather than –ic). Only choice D has the correct prefix/suffix combination.
Since polyatomic ions frequently contain an atom combined with one or more oxygen atoms, acids made from these ions are often called oxyacids. Another reason for this name is that the acidic hydrogen is often bonded to an oxygen atom. The figure to the right shows the structure of perchloric acid, HClO4, which is a typical oxyacid.

For example, HNO2 is made from hydrogen ion and the nitrite ion, NO21−. Following the rule above turns nitrite into nitrous acid. Now consider the oxyacid of nitrogen that has one additional oxygen atom, HNO3. It is made from hydrogen ion and the nitrate ion. Using the rule above turns nitrate into nitric acid.
Remember that some polyatomic ions have a prefix of hypo or per. These prefixes are not affected by the rules we have just learned. For example, HClO is made from a hydrogen ion and the hypochlorite ion, ClO1−. We still simply change –ite to –ous acid, which yields the name hypochlorous acid. The prefix hypo is retained in the name.
Which of the following correctly names the anion present in iodic acid?
The correct answer is C. An ending of “-ic acid” (without a prefix of “hydro”) means that the anion ends with “-ate.” The acid formed from choice A would be hydroiodic acid and from choice B would be iodous acid. Choice D is not an anion at all, so it cannot form an acid as such.
You may recall that metals bond with nonmetals ionically — that is, they transfer one or more electrons from the metal to the nonmetal, creating charged particles that are electrostatically attracted to each other. Because oxygen is a nonmetal, it follows logically that metal oxides are ionic compounds.
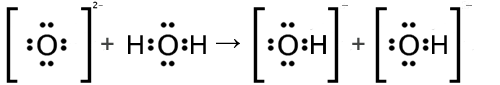
The oxide ion, O2−, behaves as a relatively strong Bronsted-Lowry base by removing a hydrogen ion from a water molecule. When this occurs, the products are two hydroxide ions, OH1− as seen in the Lewis structures below.
Which of the following correctly shows the product formed when calcium oxide reacts with water?
The correct answer is D. The oxide ion will combine with water to form hydroxide ion, ruling out choices B and C. Since calcium forms 2+ ions and hydroxide ions are 1−, there must be two hydroxide ions for every one calcium ion, as in choice D.
When oxygen bonds with a nonmetal, it does so covalently, therefore the compound does not contain oxide ions. That’s why adding water does not form hydroxide ions. Instead, the oxygen atom tends to bond with the other nonmetal in the compound, while the hydrogen atoms bond to the oxygen atoms. This is precisely the structure of an oxyacid, as discussed earlier.
Since the hydrogen atoms are not reduced to hydrogen gas (H2) and the oxygen atoms are not oxidized to oxygen gas (O2), the nonmetal originally bonded to the oxygen must retain its original oxidation number. For example, in the reaction below, dichlorine monoxide (Cl2O) reacts with water to form hypochlorous acid. The oxidation number of chlorine is +1 in both compounds.
Cl2O + H2O → 2HClO
Which of the following correctly shows the product formed when SO2 reacts with water?
The correct answer is B. SO2 is a nonmetal oxide, so it will form an acid, ruling out choice D. The oxidation number of sulfur in SO2 is +4, which is the same as its oxidation number in H2SO3. Choices A and C are incorrect as the oxidation numbers for sulfur are +2 and +6, respectively.
Which of the following correctly shows the product formed when dinitrogen pentoxide, N2O5, reacts with water?
Choice C is correct. N2O5 is a nonmetal oxide, so it will form an oxyacid with water, ruling out choices A and D. The oxidation numbers of nitrogen in choices B and C are +3 and + 5, respectively. Only choice C matches the oxidation number of nitrogen in N2O5.
Which of the following correctly shows the product formed when potassium oxide, K2O, reacts with water?
Choice A is correct. Metal oxides react with water to form metal hydroxides, eliminating choices C and D. Choice B does not have charges that add up to zero, so its formula is incorrect..
Which of the following correctly shows the product formed when tetraphosphorus decoxide, P4O10, reacts with water?
Choice D is correct. The nonmetal oxide reacts with water to form an oxyacid. The oxidation number of phosphorus in the oxyacid must be the same as its oxidation number in the original oxide, +5. Only choice D has a ratio of ions whose charges add up to zero and has an oxidation number for phosphorus of +5.